 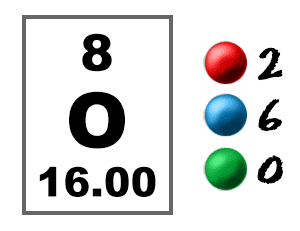 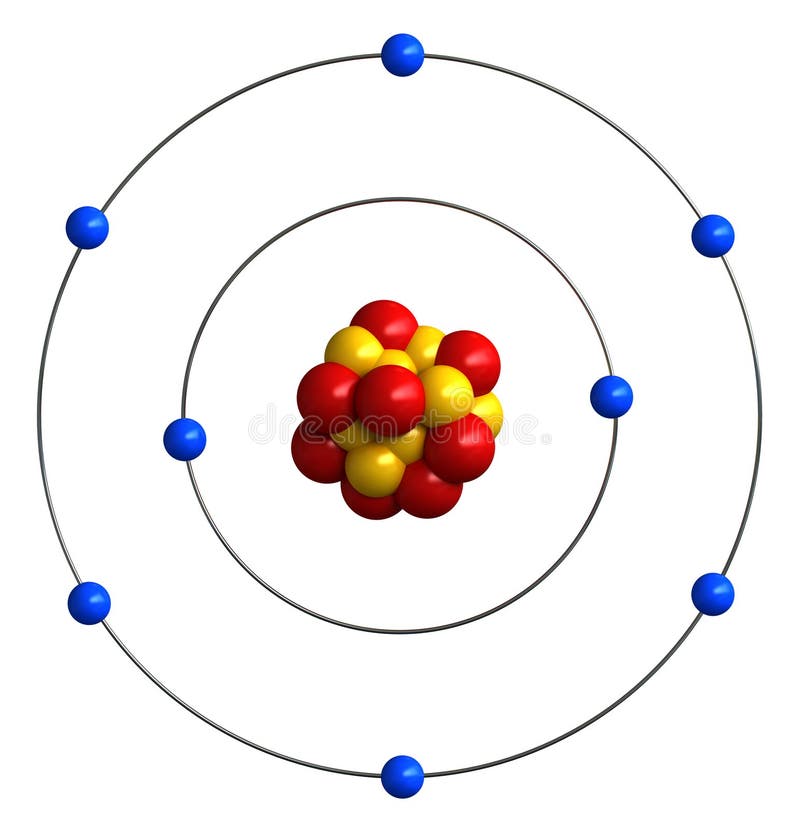
The view of oxidation and reduction as the loss and gain of electrons, respectively, is particularly appropriate for discussing reactions in electrochemical cells. Treating that charge as an " oxidation number" is another way to characterize oxidation and reduction. Another way to judge that the chlorine has been reduced is the fact that the charge on the atoms is made more negative, or reduced. Magnesium loses electrons and is therefore said to be "oxidized", whereas the chlorines gain electrons and are said to be reduced. For example, the reaction Mg + Cl 2 -> Mg 2+ + 2Cl. This electron view of oxidation and reduction helps you deal with the fact that "oxidation" can occur even when there is no oxygen! The definition of redox reactions is extended to include other reactions with nonmetals such as chlorine and bromine. In this reaction the lead atoms gain an electron (reduction) while the oxygen loses electrons (oxidation). One example in which this approach is of value is in the high temperature reaction of lead dioxide. An alternative view is to describe oxidation as the losing of electrons and reduction as the gaining of electrons. 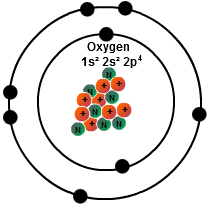
The original view of oxidation and reduction is that of adding or removing oxygen. The CO is reduced because it gains hydrogen, and the hydrogen is oxidized by its association with the oxygen. The formation of methanol from reacting carbon monoxide with hydrogen combines oxidation and reduction in the single molecular product. 
The formation of H 2 is a reduction process as the two released hydrogens get together. Another reaction where the hydrogen approach makes things clearer is the passing of methanol over a hot copper gauze to form formaldehyde and hydrogen gas (Hill and Kolb): CH 3OH -> CH 2O + H 2īoth carbon-containing molecules have the same oxygen content, but the formation of the formaldehyde is seen to be oxidation because hydrogens are lost. With this approach it is clear that the carbon is oxidized (loses all four hydrogens) and that part of the oxygen is reduced (gains hydrogen). This has an advantage in describing the burning of methane. An alternative approach is to describe oxidation as the loss of hydrogen and reduction as the gaining of hydrogen. HyperPhysics***** Quantum Physics ***** Chemistry The reduction of the lead dioxide is clear, but the associated oxidation of oxygen is easier to see when you describe oxidation as the losing of electrons. On the other hand, the reaction of lead dioxide at high temperatures appears to be just reduction. The accompanying reduction of oxygen is perhaps easier to see when you describe reduction as the gaining of hydrogen. For example in the burning of methane, CH 4 + 2O 2 -> CO 2 + 2H 2Oīoth carbon and hydrogen are oxidized (gain oxygen). In some reactions, the oxidation is most prominent. This formation of nitric oxide oxidizes the nitrogen and reduces the oxygen. The combination of nitrogen and oxygen which occurs at high temperatures follows the same pattern. The hydrogen is oxidized and the oxygen is reduced. For example, in the burning of hydrogen 2H 2 + O 2 -> 2H 2O The earliest view of oxidation and reduction is that of adding oxygen to form an oxide (oxidation) or removing oxygen (reduction). Oxidation and Reduction Gain and Loss of Oxygen 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
